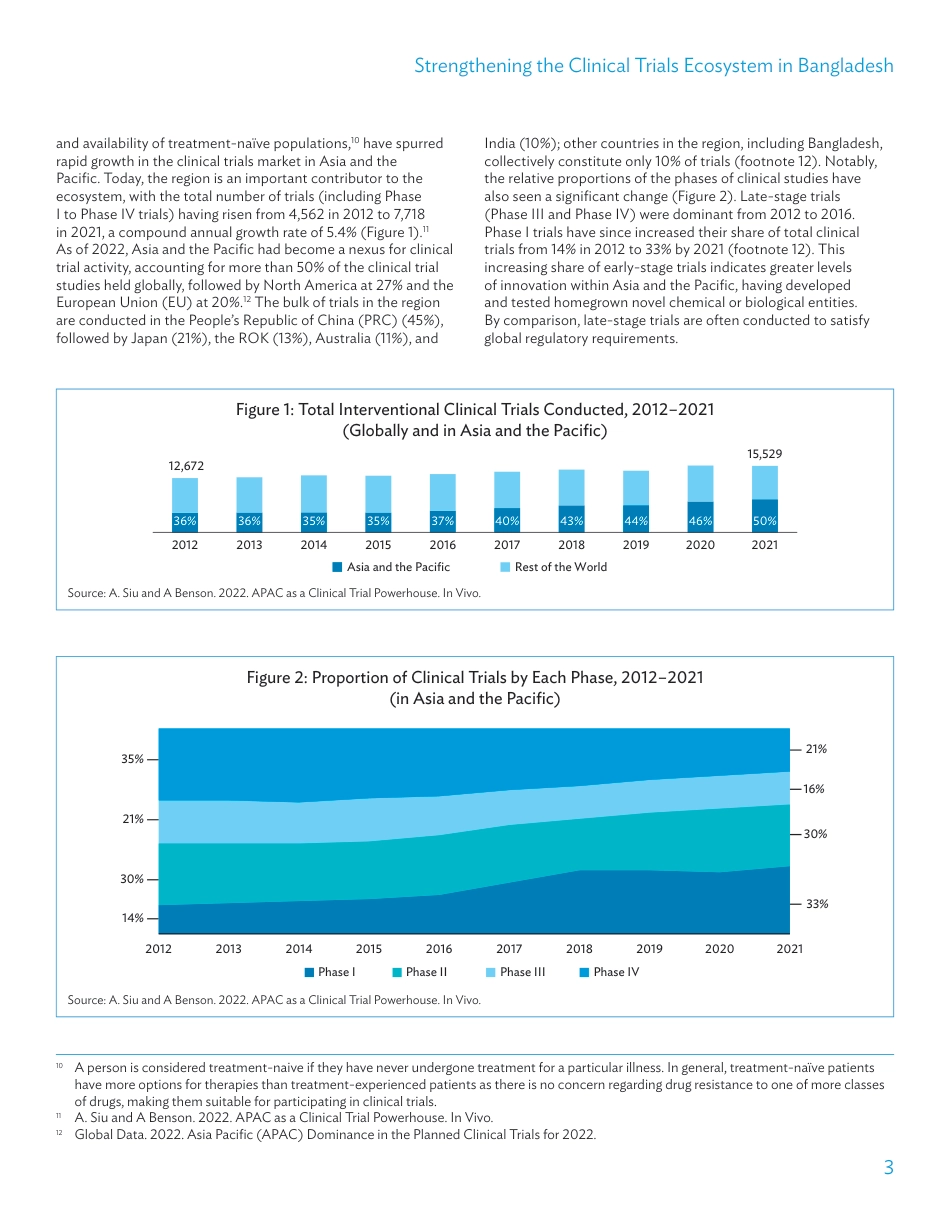
ADBBRIEFSNO.308JULY2024StrengtheningtheClinicalTrialsEcosysteminBangladesh:LandscapeandPolicyRecommendationsKEYPOINTS•Awell-functioningclinicaltrialsecosystemisessentialtostrengtheningdomesticpharmaceuticalmanufacturinginlow-andmiddle-incomecountries,aslocalresearchanddevelopmentcancontributetoself-sufficiencyandsupplysecurityforvaccinesandtherapeutics.•AsiaandthePacifichasbecomeanexusforclinicaltrialactivityoverthepastdecade,becauseoffactorssuchasloweroperationalcosts,rapidlyincreasingcapabilities,andlargepotentialmarketsizes.•WhiletherearesomeclinicaltrialsitesthatareexperiencedinconductingtrialsforinfectiousdiseasespresentinBangladesh,overallactivityhasremainedrelativelylowandconcentratedinafewareas,withlowindustryparticipation.•Policyactionssuchasregulatoryprocessoverhaul,strengtheningofclinicaltrialinfrastructure,capacity-buildinginitiatives,prioritizationofdataprivacyandtransparency,andfinancialincentivescanhelpbuildtheenablingconditionsrequiredforastrongclinicaltrialsecosysteminBangladesh.ISBN978-92-9270-799-6(print)ISBN978-92-9270-800-9(PDF)ISSN2071-7202(print)ISSN2218-2675(PDF)PublicationStockNo.BRF240355-2DOI:http://dx.doi.org/10.22617/BRF240355-2INTRODUCTIONANDBACKGROUNDThecoronavirusdisease(COVID-19)pandemichighlightedhowpublichealthemergenciescanhavesevereimpactsontheoverallhealthsecurityofco...












发表评论取消回复